Tag: ANDA approval
Bioequivalence Studies: What the FDA Requires Manufacturers to Prove
- Laura Ledas
- Feb, 15 2026
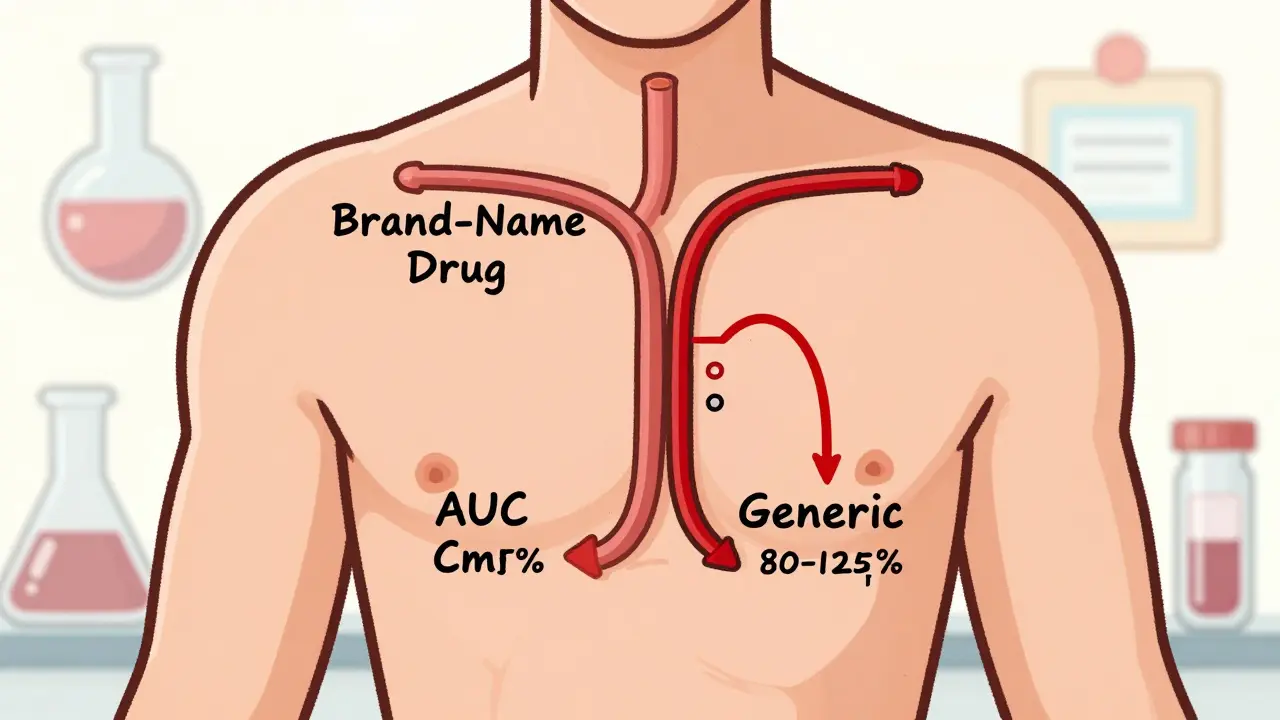
The FDA requires generic drug manufacturers to prove bioequivalence through rigorous clinical studies that show their product matches the brand-name drug in absorption and blood concentration. This ensures safety, effectiveness, and therapeutic equivalence.
Learn MoreGeneric Drug Quality Standards: FDA Requirements and Testing
- Laura Ledas
- Jan, 31 2026
Generic drugs must meet the same strict FDA standards as brand-name drugs, including pharmaceutical and bioequivalence. Learn how the FDA ensures quality, safety, and effectiveness through rigorous testing, inspections, and approval processes.
Learn More